Adsorption rate increases as the surface area increases.Physical adsorption is facilitated by low temperatures, while high temperatures slow the adsorption process. So, lowering the pressure removes gas from a solid surface. The volume of gas drops due to a rise in pressure, and more gas is adsorbable. In nature, physical adsorption is a reversible process.Easily liquefiable gases are physically adsorbed to a great extent.Each gas is adsorbed onto the solid’s surface. In the event of physical adsorption, there is no specialisation.Van der Waals forces play a role in the adsorption of gases onto a solid surface. Due to their high surface area, activated charcoal, silica, and alumina, among others, are extremely effective adsorbents.Īdsorption is classified into two types – physical adsorption, also known as physisorption, and chemical adsorption, also known as chemisorption. It includes metal powders, charcoal powder, and silica powder. Adsorbent: When adsorption occurs, the substance that forms a surface on which it can take place is referred to as adsorbent.For example, if gas is adsorbed on the solid surface, it is referred to as the adsorbate. Adsorbate: Adsorbate is the term used to describe the adsorbed substance on a surface.This surface phenomenon requires two components – adsorbate and adsorbent. Surface chemistry is concerned with the events at or near surfaces or interfaces. It is typically composed of dissolved gas, liquids, solid molecules, atoms, or ions. When it comes to surface phenomena, adsorption is the adhesion of particles to the surface. 
Heinrich Kayser, a German scientist, coined the term adsorption in 1881. AdsorptionĪdsorption is the phenomenon through which atoms, molecules, or ionic constituents of one substance aggregate on the surface. Attracting and keeping particles on the liquid or solid surface causes the molecules to become more concentrated on that surface. 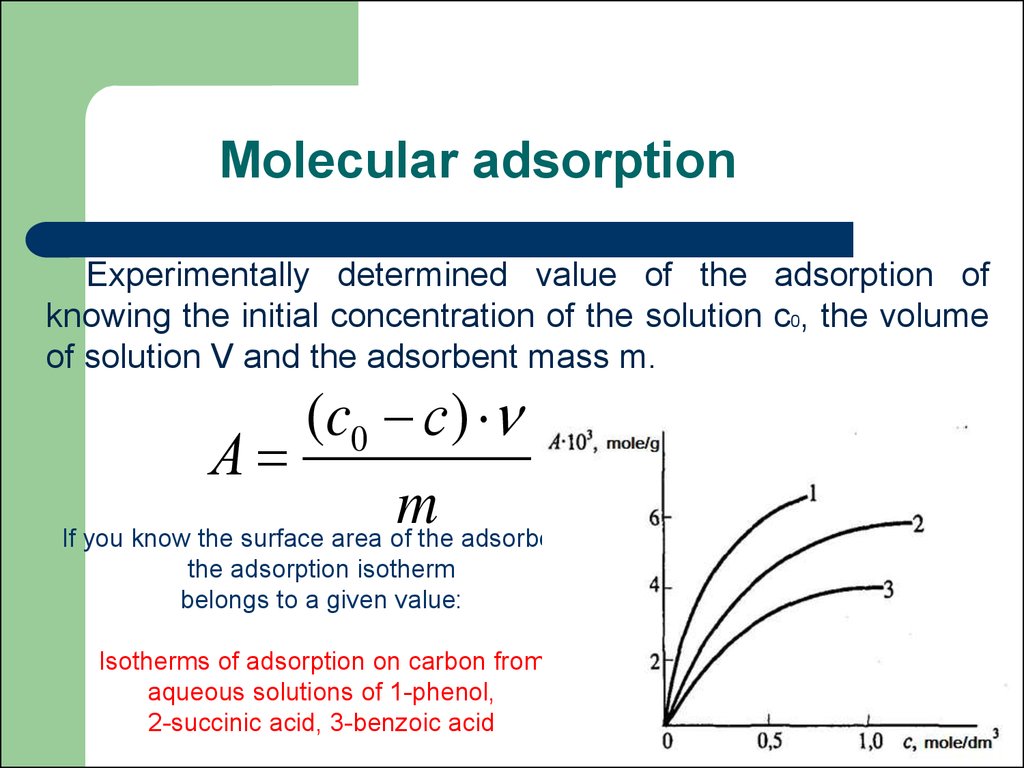
Adsorption, like surface tension, results from the presence of surface energy. It’s important to note that absorption occurs across the entire volume, whereas adsorption appears on the surface. Adsorption is the process by which atoms, ions, or molecules attach to a surface from a gas, liquid, or dissolved solids.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
